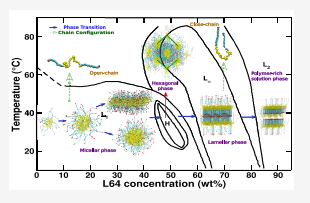
Understanding the self-assembly of amphiphilic molecules in supercooled solvents using molecular simulations
The self-assembly in an aqueous medium is a major scientific area for many practical and biological applications, in which freezing induced self-assembly (FISA) is considered to be a more novel method as compared to solvent evaporation induced self-assembly. FISA is a versatile and green bottom-up method for producing highly ordered and aligned porous materials. The molecular details of these processes are largely unknown because of the very small length scale involved (nanometres). We have attempted to use seeding methodology to capture the nucleation behaviour of water in the presence of amphiphilic molecules. The understanding has been previously utilized for stationary fluids and sheared flows. However, the underneath algorithm is quite valuable for the current project as well. Crystal nucleation is a rare event that can occur on time scales of seconds, far beyond the reach of the brute-force molecular dynamics framework. Seeding overcomes this barrier by the insertion of ice seed in a pre-equilibrated solution. If the ice seed is larger than the critical nucleus, it will grow with time evolution, leading to the formation of different ice crystals and self-assembly of amphiphilic molecules into 3D or 2D structures. Our work provides preliminary molecular insights into the factors affecting the nucleating behaviour of water in the presence of amphiphilic molecules, which play a pivotal role in designing artificial ice recrystallization inhibition (IRI) agents, for use in cryobiology and drug delivery.
|